|
While rare stromal cells will be indicated, they are not considered consistent with a diagnosis of endometritis.Biopsy samples taken from your esophagus (typically during an endoscopy) are studied by a doctor with special training, called a pathologist. The results are calculated and reported based on presence of clusters of plasma cells. For clinical purposes, a value of 1.5 vs 4 will not affect protocols.ĬD138 is an antigen specific to plasma cells and used to detect chronic endometritis. Any results above 1.4 should be considered positive and treated according to the guidelines under treatment. The use of the test specific to infertility has been validated against laparoscopy-confirmed endometriosis patients using a cutoff value of 1.4. Please note that the H-Score value is not an indicator of endometriosis disease stage. The H-score is calculated as the sum of the percentage of staining multiplied by an ordinal value corresponding to the intensity level and reported as (0 = none, 1 = weak, 2 = moderate, 3 = strong 4= very strong). Results are reported as an H-score between 1-4. Please call 80 to register your center.īoth biomarkers BCL6 and CD138 are performed using Immunohistochemistry.īCL6 is a protein marker highly associated with endometriosis. Results can only be faxed to fax number provided on test request form. Results will be available 5-6 business days from date received by lab. NOTE: sample is stable at room temperature. Preferred shipment days are Monday-Friday FedEx overnight. 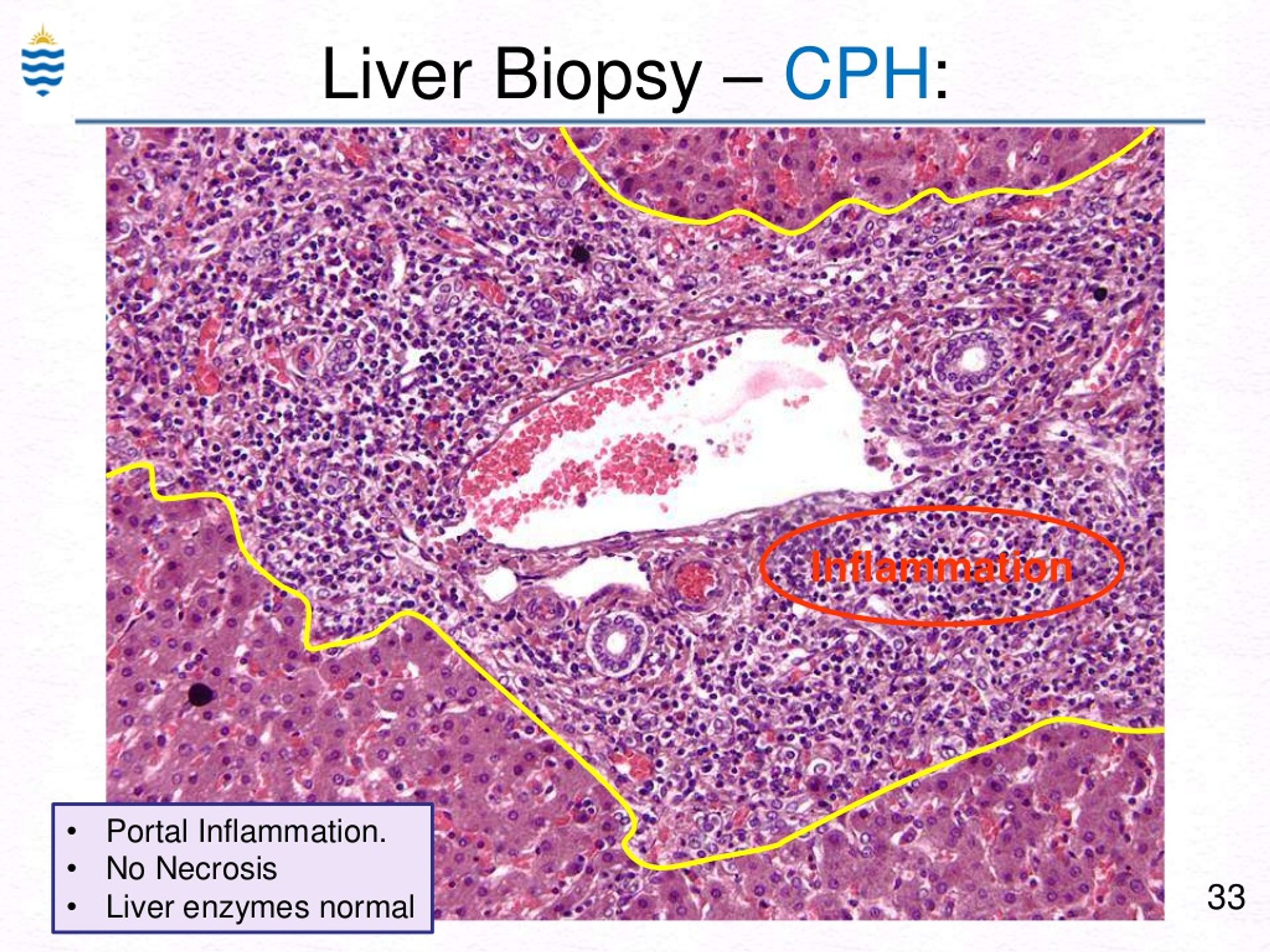
Place bag and test request form in ReceptivaDx Collection box.Verify test request form is complete, including payment information and patient email.Verify two patient identifiers on vial match information on test request form.Place vial in hazard/safety bag (provided in collection kit).Make sure vial cap is secured (clicking sound of cap indicates sample is secure).NOTE: Do not use vial provided by other companies. Samples are collected in formalin only (provided in collection kit).Cicero Diagnostics has performed more than 20,000 tests and continues to collect and publish results on outcomes. Since that early paper, over a dozen additional publications have come out highlighting the effectiveness of the test. Results were successful in predicting the likelihood of live birth on the next IVF transfer for patients experiencing unexplained infertility prior to the transfer. A December 2017 cohort study in Fertility and Sterility demonstrated the prognostic value of BCL6 results. A negative BCL6 result provides reassurance to patients that endometriosis is not of concern. 
Studies have shown that women with elevated BCL6 results are 5 times less likely to succeed in IVF or subsequent transfer attempts. BCL6 is dramatically over-expressed in women with endometriosis, which makes it a likely candidate for the cause of progesterone resistance that contributes to implantation problems (read blog post). Progesterone is required for a normal pregnancy, so a protein that interferes with progesterone action would explain much about the problems encountered in women with endometriosis. The test specifically identifies BCL6, a protein biomarker associated with inflammation. The results can help indicate what is likely causing implantation issues. While other tests evaluate the quality of embryos or determine optimal embryo transfer windows, only ReceptivaDx can accurately detect inflammatory conditions on the uterine lining.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
